RSV infects cells along the respiratory tract from the nose to the lungs, causing a wide spectrum of respiratory disease, including lower respiratory tract infection1


Nasal passage
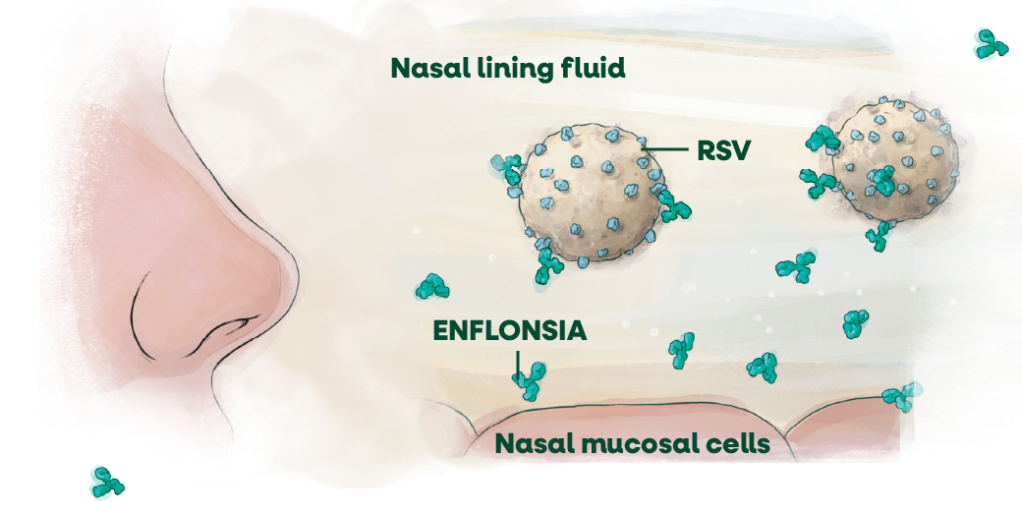

ENFLONSIA has been detected in the lining fluid of the nasal mucosal cells in adults—an important point of viral entry into the body.2,3
RSV-neutralizing antibody titers in serum were estimated to be ~7x higher than baseline levels at 4 hours following administration
RSV membrane: a closer look
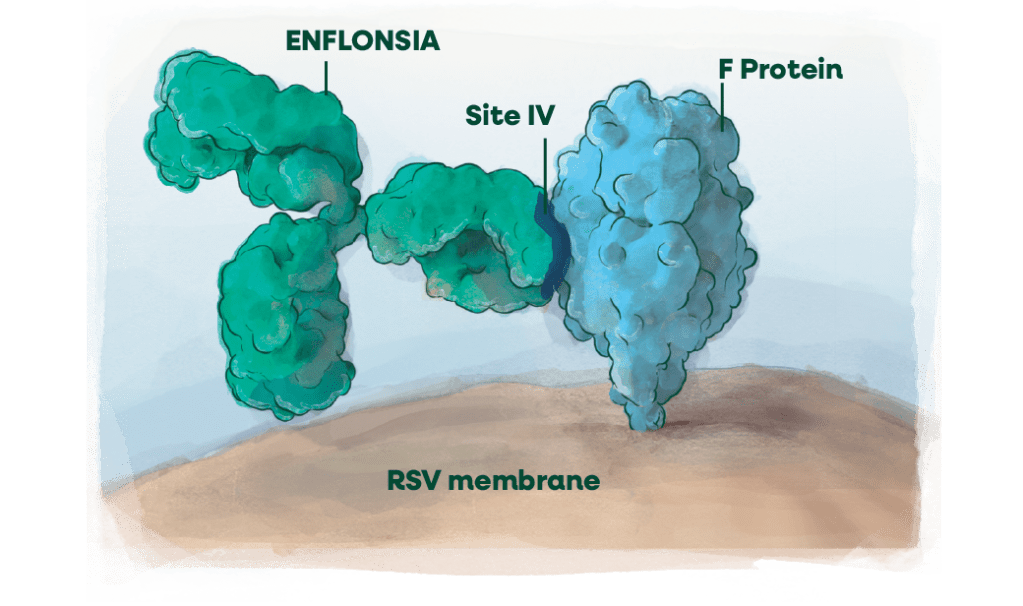

- Binds to antigenic site IV (highly conserved in 99.8% of strains)a
- Fully human mAb modified for extended serum half-lifeb
aIn sequences reported in GenBank database (accessed 4/2024).
bModified with a triple amino acid substitution (YTE) in the Fc region.
F protein, fusion protein; IM, intramuscular; RSV, respiratory syncytial virus; Fc, Fragment crystallizable.
References:
- Respiratory syncytial virus (RSV). World Health Organization. Published December 19, 2025. Accessed January 6, 2025. https://www.who.int/news-room/fact-sheets/detail/respiratory-syncytial-virus-(rsv)#
- How RSV spreads. Centers for Disease Control and Prevention. July 8, 2025. Accessed July 24, 2025. https://www.cdc.gov/rsv/causes/
- Phuah JY, Maas BM, Tang A, et al. Quantification of clesrovimab, an investigational, half-life extended, anti-respiratory syncytial virus protein F human monoclonal antibody in the nasal epithelial lining fluid of healthy adults. Biomed Pharmacother. 2023;169:115851. doi:10.1016/j.biopha.2023.115851
