ENFLONSIA is available for ordering as a single-dose prefilled syringe in either one or ten pack of 0.7 mL syringes for commercially insured patients.
ENFLONSIA is available to order via the Vaccines for Children (VFC) Program in most states. Check with your local/state program manager for more information.
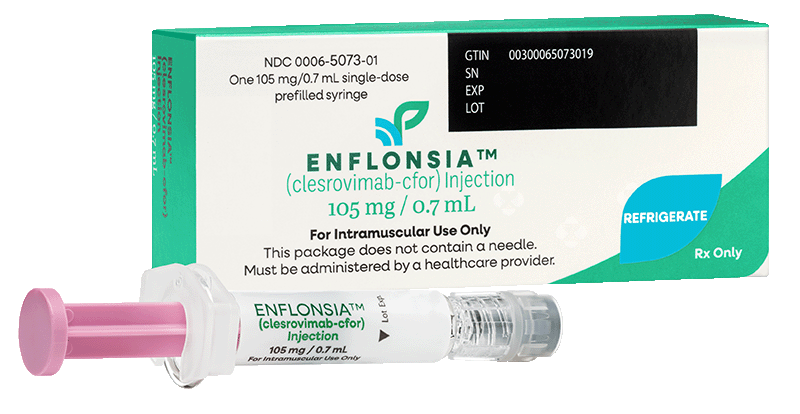
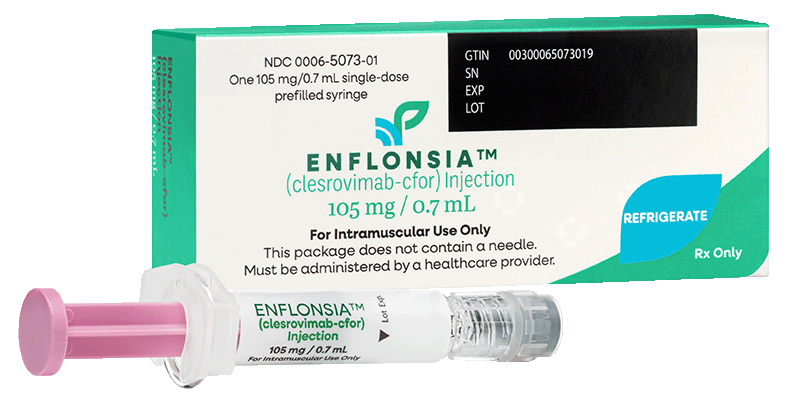
Package dimensions: Pack of 1: 44 x 105 x 29 mm
Not actual size shown
Available to order as a single-dose prefilled syringe based on the needs of your practice


Package dimensions: Pack of 10: 105 x 133 x 41 mm
Not actual size shown
ENFLONSIA is also available in a carton of ten single-prefilled Luer Lock syringes with tip caps, each syringe containing 1 dose of 105 mg/0.7 mL
Supply options
ENFLONSIA injection is a sterile, preservative-free, clear to slightly opalescent, colorless to slightly yellow solution supplied as follows:
- Carton containing one or ten single-dose prefilled type I glass syringe(s) with Luer Lock and plunger stopper. The prefilled syringe is not made with natural rubber latex.
| Prefilled syringe | Pack size | NDC |
|---|---|---|
| 105 mg/0.7 mL single-dose | Pack of 1 | 10-Digit: 0006-5073-01 11-Digit: 00006-5073-01 |
| 105 mg/0.7 mL single-dose | Pack of 10 | 10-Digit: 0006-5073-02 11-Digit: 00006-5073-02 |
NDC, National Drug Code
