Purchased ENFLONSIA doses may be used for the next RSV season for infants entering their first RSV season.
Before storing for next season, it is important to check the expiration date to ensure the product won’t expire during off season storage.
Storage & handling
Single doses available!
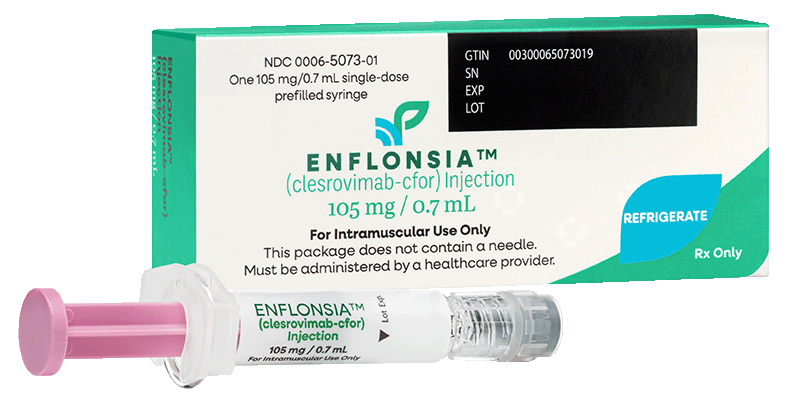

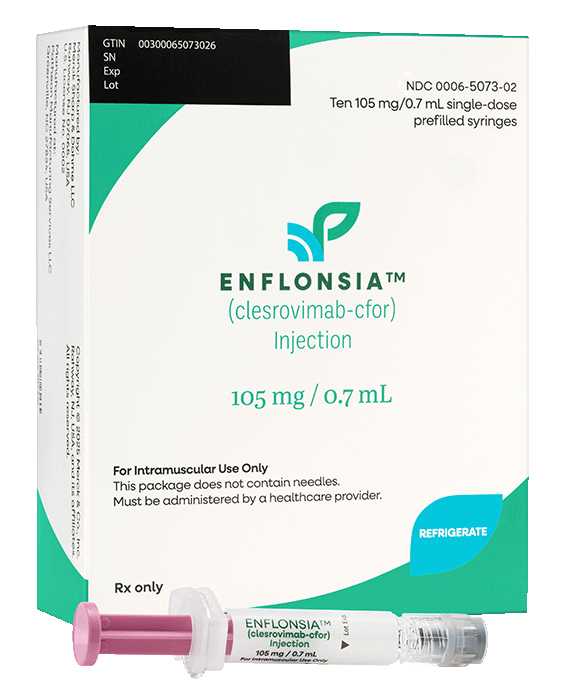

Not actual size shown
Share with health care professionals
Reference:
- US Food & Drug Administration Center for Drug Evaluation and Research. BLA Approval Letter. Published June 9, 2025. Accessed October 21, 2025. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2025/761432Orig1s000Approv.pdf
