Do not administer VAQTA to individuals with a history of immediate and/or severe allergic or hypersensitivity reactions (eg, anaphylaxis) after a previous dose of any hepatitis A vaccine, or to individuals who have had an anaphylactic reaction to any component of VAQTA, including neomycin.
The vial stopper contains dry natural latex rubber that may cause allergic reactions in latex- sensitive individuals.
The most common local adverse reactions and systemic adverse events (≥15%) reported in different clinical trials across different age groups when VAQTA was administered alone or concomitantly were:
- Adults 19 years of age and older: injection-site pain, tenderness, or soreness (67.0%), injection site warmth (18.2%), and headache (16.1%).
Hepatitis A virus has a relatively long incubation period (approximately 20 to 50 days). VAQTA may not prevent hepatitis A infection in individuals who have an unrecognized hepatitis A infection at the time of vaccination.
Immunocompromised persons, including individuals receiving immunosuppressive therapy, may have a diminished immune response to VAQTA and may not be protected against HAV infection after vaccination.
Vaccination with VAQTA may not result in a protective response in all susceptible vaccinees.
VAQTA may be administered concomitantly with Immune Globulin, human, using separate sites and syringes.
There are no adequate and well-controlled studies designed to evaluate VAQTA in pregnant women, including those 19 years of age or younger. Available post-approval data do not suggest an increased risk of miscarriage or major birth defects in women who received VAQTA during pregnancy.
It is not known whether VAQTA is excreted in human milk. Data are not available to assess the effects of VAQTA on the breastfed infant or on milk production/excretion. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VAQTA and any potential adverse effects on the breastfed child from VAQTA or from the underlying maternal condition.
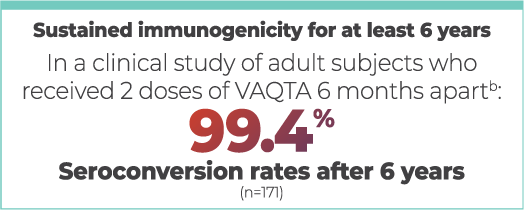
The total duration of the protective effect of VAQTA in healthy vaccinees is unknown at present.
In clinical trials in adults, VAQTA was concomitantly administered with typhoid Vi polysaccharide and yellow fever vaccines. Safety and immunogenicity were similar for concomitantly administered vaccines compared to separately administered vaccines.
Dosage and Administration
Adults (19 years of age and older): The vaccination schedule consists of a primary 1 mL dose administered intramuscularly and a 1 mL booster dose administered intramuscularly 6 to 18 months later.
Booster Immunization Following Another Manufacturer’s Hepatitis A Vaccine: A booster dose of VAQTA may be given at 6 to 12 months following a primary dose of Havrix*.
*Havrix is a registered trademark of GSK.
Before administering VAQTA, please read the accompanying Prescribing Information. The Patient Information also is available.