
Pediatric storage and handling for VAQTA® (Hepatitis A Vaccine, Inactivated)
Condition on arrival
Should not be frozen. Refrigerate on arrival.1
Storage
Store vaccine at 2°C to 8°C (36°F to 46°F). DO NOT FREEZE since freezing destroys potency.
How supplied
VAQTA is available in prefilled Luer-Lok® syringes.
Pediatric/Adolescent formulations
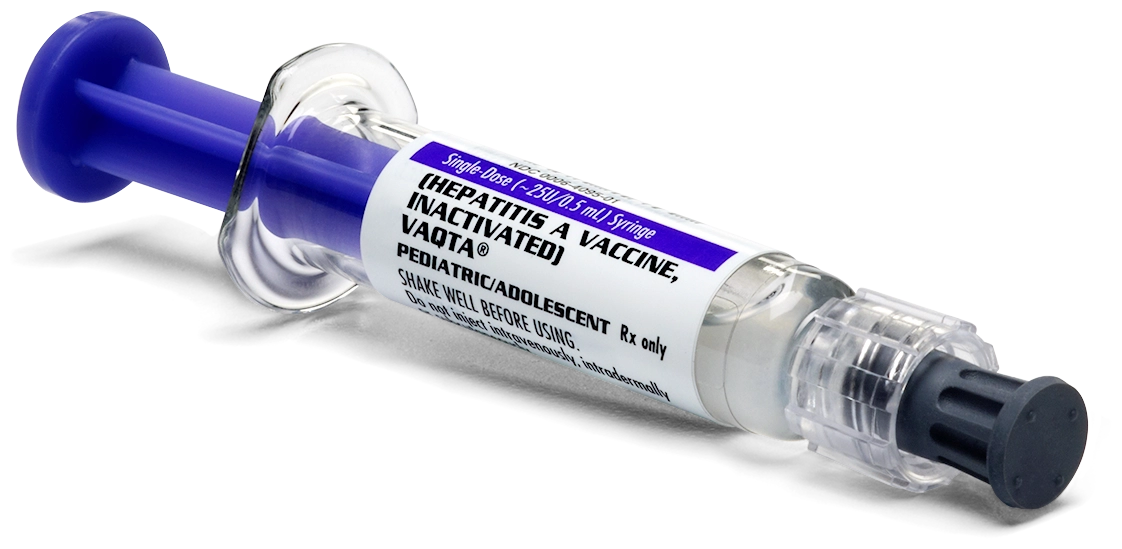
Not shown actual size
NDC 0006-4095-02
Brands mentioned are the trademarks of their respective owners.
General tips
- If you have questions about the condition of the vaccine at the time of delivery, you should immediately place vaccine in recommended storage and call the Merck Vaccine Customer Center at 877.VAX.MERCK (877.829.6372).
- Rotate stock so that doses with the earliest expiration dates are used first.1
- Ensure that the refrigerator is plugged into an outlet in a protected area where it cannot be disconnected accidentally.1
- Vaccines For Children (VFC) Program providers should separate and identify VFC and other vaccines purchased with public funds within the storage unit. In addition, clearly label the space where the vaccine is placed to help staff choose the appropriate vaccine.1
- A separate needle and syringe should be used for each injection. Changing needles between drawing vaccine from a vial and injecting it into a recipient is not necessary unless the needle has been damaged or contaminated.2,3
- For general questions concerning the proper storage and handling of Merck vaccines, please contact the Merck Vaccine Customer Helpline at 800.MERCK.90 (800.637.2590), Monday through Friday, 8:00 AM to 7:00 PM ET.
References
- Centers for Disease Control and Prevention (CDC). Vaccine Storage and Handling Toolkit. Last reviewed March 29, 2024. Accessed January 15, 2026. https://www.cdc.gov/vaccines/hcp/admin/storage/toolkit/storage-handling-toolkit.pdf
- Centers for Disease Control and Prevention (CDC). Vaccine Administration. United States, Last updated June 18, 2024. Accessed January, 2026. https://www.cdc.gov/vaccines/hcp/imz-best-practices/vaccine-administration.html
- Centers for Disease Control and Prevention. Vaccination programs. CDC Vaccines & Immunizations. Updated July 17, 2024. Accessed January 15, 2026. https://www.cdc.gov/vaccines/hcp/imz-best-practices/vaccination-programs.html#
