
VAQTA® (Hepatitis A Vaccine, Inactivated) pediatric clinical data – efficacy and immunogenicity
VAQTA has been FDA-approved for over 25 years.
VAQTA demonstrated 100% efficacy against hepatitis A in the landmark Monroe study of 2 to 16-year-oldsa




In the Monroe study, VAQTA demonstrated 100% efficacy against hepatitis A with 21 cases occurring in the placebo group and none in the vaccine group (P < 0.001) ≥50 days after vaccination.
Following review of the interim analysis of the initial study after a single dose, the monitoring committee recommended that the study be terminated so that the placebo recipients could be vaccinated. A booster dose was administered to a subset of vaccinees 6, 12, or 18 months after the primary dose.
Clinical study design
aAn initial randomized, double-blind, placebo-controlled study was conducted to evaluate the efficacy of a single dose of VAQTA in a community with recurrent outbreaks. In this study, 1037 children, 2 to 16 years of age, were enrolled. Each child received an intramuscular dose of VAQTA (25 U) (n=519) or placebo (n=518). Efficacy was based on clinically confirmed cases of hepatitis A occurring ≥50 days after vaccination.
After 5 months, the initial study was terminated so that the vaccine could be provided to the subjects in the placebo group. A second (booster) dose was administered at 6, 12, or 18 months after the first dose of VAQTA. All vaccines were closely monitored for clinically confirmed cases of hepatitis A from 1991 to January 2001.
The total duration of the protective effect of VAQTA in healthy vaccinees is unknown at present.
Vaccination with VAQTA may not result in a protective response in all susceptible vaccinees.
VAQTA demonstrated 100% efficacy*across multiple studies after the second dose.
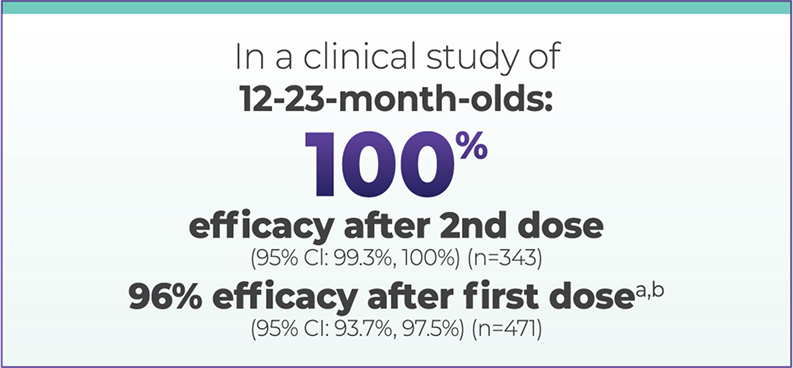



Clinical study design
The efficacy of VAQTA was evaluated in a clinical trial that included children 12 through 23 months of age who were randomized to receive the first dose of VAQTA with or without M-M-R®II (Measles, Mumps, and Rubella Virus Vaccine Live) and VARIVAX® (Varicella Virus Vaccine Live) and the second dose of VAQTA with or without Tripedia (diphtheria and tetanus toxoids and acellular pertussis vaccine) and optionally either oral poliovirus vaccine (no longer licensed in the US) or IPOL (Poliovirus Vaccine Inactivated).
Immunogenicity data were combined from 11 randomized clinical studies in children and adolescents 2 through 18 years of age who received VAQTA (25 U/0.5 mL). These included administration of VAQTA in varying doses and regimens (N=404 received 25 U/0.5 mL), the Monroe Efficacy Study (N=973), and comparison studies for process and formulation changes (N=1238).
*The efficacy of VAQTA in 1 through 18 year-olds was based upon immunogenicity measured 4 to 6 weeks following vaccination.
- Vaccination with VAQTA may not result in a protective response in all susceptible vaccinees
- The total duration of the protective effect of VAQTA in healthy vaccinees is unknown at present
aIn initially seronegative children who received VAQTA with or without other vaccines.
bPost dose 1: anti-HAV GMT = 48 mIU/mL (95% CI: 44.7, 51.6). Post dose 2: anti-HAV GMT = 6920 mIU/mL (95% CI: 6136, 7801).
cPost dose 1: anti-HAV geometric mean titer (GMT) = 43 mIU/mL (95% CI: 40, 45). Post dose 2: anti-HAV GMT = 10,077 mIU/mL (95% CI: 9394, 10,810).
CI, confidence interval; GMT, geometric mean titer; HAV, hepatitis A virus.
There are many opportunities to vaccinate your pediatric patients1
Reference
- American Academy of Pediatrics. The American Academy of Pediatrics’ recommended child and adolescent immunization schedule. Updated February 5, 2026. Accessed February 12, 2026. https://publications.aap.org/redbook/resources/15585/