Measles outbreaks can still occur within the US.3,4
~1 out of 10 children were unvaccinated against MMR by 24 months of age4,5,a
aNIS-Child is a random-digit-dialed survey of households that included children aged 19–35 months. Parents or guardians completed a telephone survey and consent to contact the child’s vaccination providers was requested. With parental or guardian consent, identified providers were mailed a questionnaire to obtain vaccination information, which was synthesized to create the child’s comprehensive vaccination history. Children born during 2020–2021 were identified from data collected during 2021–2023, resulting in 28,688 children with adequate provider data for analysis.
Remember to check for children and adolescents who are unvaccinated against measles, mumps, and rubella viruses3,4
The CDC recommends that children complete a 2-dose vaccine series against MMR viruses.1
M-M-R®II can be used for both doses1,6

1st Dose
12-15 months of age

2nd Dosea
4-6 years of age
aSecond dose can be administered as early as 4 weeks after the first dose.1
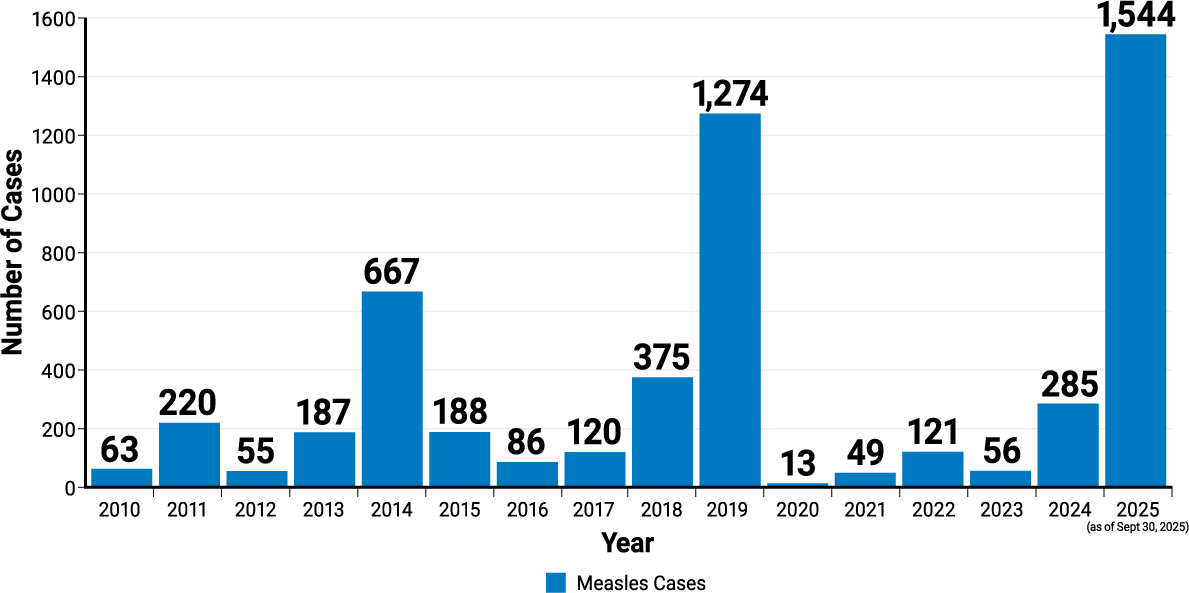
Number of measles cases reported by year3
2010-2025 (as of September 30, 2025)

M-M-R®II catch-up vaccination schedule
For children and adolescents who are unvaccinated against MMR viruses, the CDC recommends catch-up vaccination consisting of a 2-dose series with doses administered at least 4 weeks apart.1
M-M-R®II: Part of the MMRV Family

Dosage and administration
Discover the dosing regimen and administration of M-M-R®II.

Clinical data
Explore the robust coverage M-M-R®II offers your pediatric patients.
References
- Recommended child and adolescent immunization schedule for ages 18 years or younger, United States, 2025. Updated October 7, 2025. Accessed October 22, 2025. https://www.cdc.gov/vaccines/hcp/imz-schedules/downloads/child/0-18yrs-child-combined-schedule.pdf
- McLean HQ, Fiebelkorn AP, Temte JL, Wallace GS; CDC. Prevention of measles, rubella, congenital rubella syndrome, and mumps, 2013: Summary recommendations of the Advisory Committee on Immunization Practices (CDC). MMWR Recomm Rep. 2013;62(RR-04):1–34.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated October 1, 2025. Accessed October 1, 2025. https://www.cdc.gov/measles/data-research/index.html
- Ackerson B, Sy LS, Glenn SC, et al. Pediatric vaccination during the COVID-19 pandemic. Pediatrics. 2021;148(1):e2020047092. doi:10.1542/peds.2020-047092
- Hill HA, Yankey D, Elam-Evans LD, et al. Decline in Vaccination Coverage by Age 24 Months and Vaccination Inequities Among Children Born in 2020 and 2021 — National Immunization Survey-Child, United States, 2021–2023. MMWR Morb Mortal Wkly Rep. 2024;73(38):844–853. doi:10.15585/mmwr.mm7338a3
- Marin M, Broder KR, Temte JL, Snider DE, Seward JF. Use of combination measles, mumps, rubella, and varicella vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP) RR-3. Vol 59. MMWR. Centers for Disease Control and Prevention; 2010.

